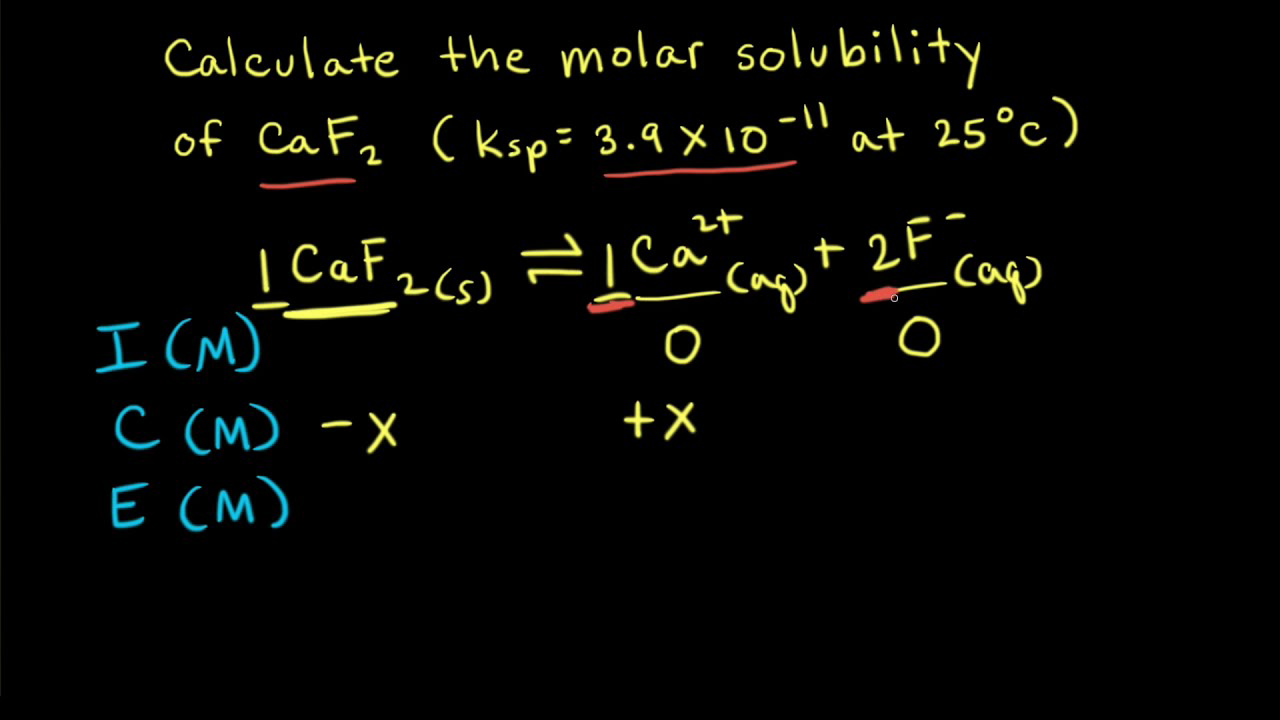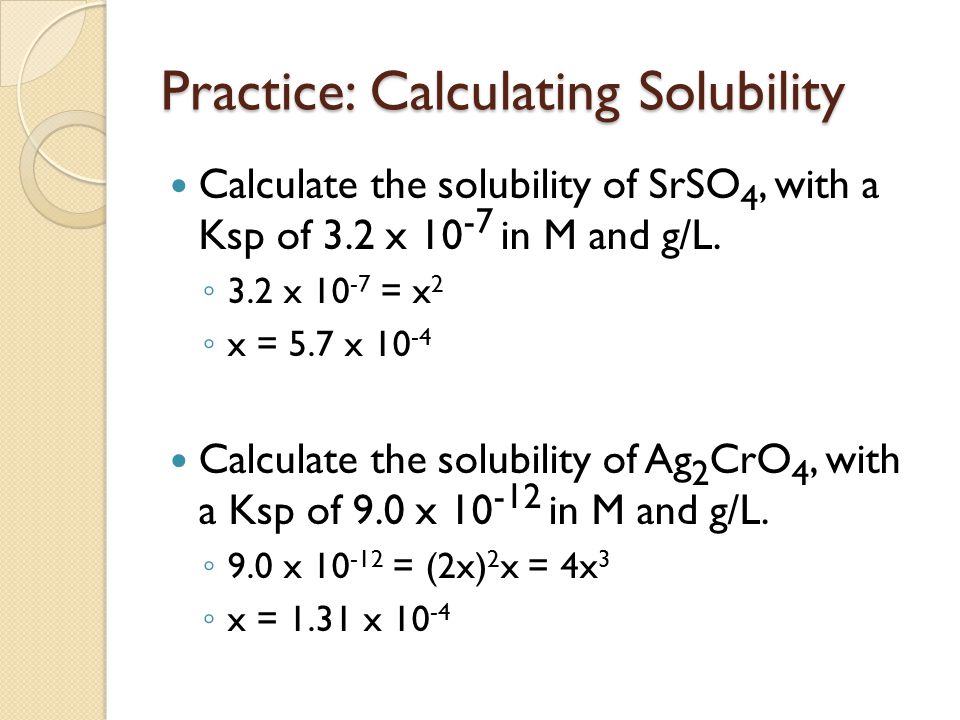the solubility of CaCO3 is 7*10^ 3 mg/litre.calculate the solubility of BaCO3 (in mol/l) from this information and from the fact that when Na2CO3 is added slowly to a solution containing equimolar
![SOLVED: #6 Calculate the total solubility of the basic drug € at pH 6.0. Additional information for drug C: SB (intrinsic solubility) = 0.8 mglmL pKa 8 2 St Sua (1+]OpH-pKa) Ans: 52 mglmL St = Sp (1+]OpKa-pH) SOLVED: #6 Calculate the total solubility of the basic drug € at pH 6.0. Additional information for drug C: SB (intrinsic solubility) = 0.8 mglmL pKa 8 2 St Sua (1+]OpH-pKa) Ans: 52 mglmL St = Sp (1+]OpKa-pH)](https://cdn.numerade.com/ask_images/7d7ca9e3ac22444fbaeef36ffd903405.jpg)
SOLVED: #6 Calculate the total solubility of the basic drug € at pH 6.0. Additional information for drug C: SB (intrinsic solubility) = 0.8 mglmL pKa 8 2 St Sua (1+]OpH-pKa) Ans: 52 mglmL St = Sp (1+]OpKa-pH)

Calculate the solubility product of PbCl(2) at a certain temperature if the solubility of PbCl(2) is 4.4 g/L at the same temperature. (Pb=207,Cl=35.5)