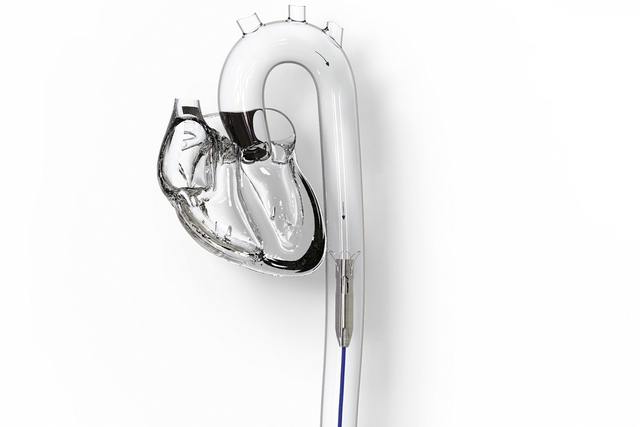
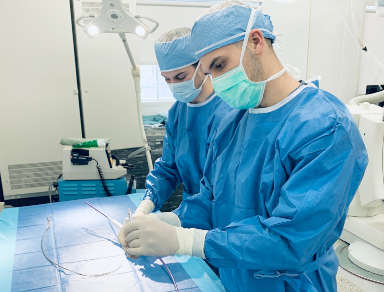
First-in-Human Experience With the ModulHeart Device for Mechanical Circulatory Support and Renal Perfusion

Puzzle Medical Closes CAN$ 34 million Series A Funding Round to Complete Human Feasibility Study Led by Cordis-X - MedTech Innovator
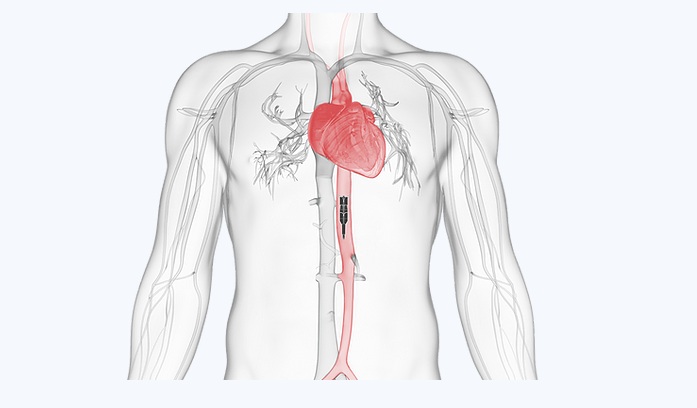
Puzzle Medical Devices Inc. Receives U.S. FDA Breakthrough Device Designation for Its Revolutionary Minimally Invasive Transcatheter Heart Pump

Finding the perfect match: The importance of chemical characterisation in medical device compatibility - Medical Plastics News

Puzzle Medical solves FDA approval process with breakthrough device designation | 2021-02-19 | BioWorld