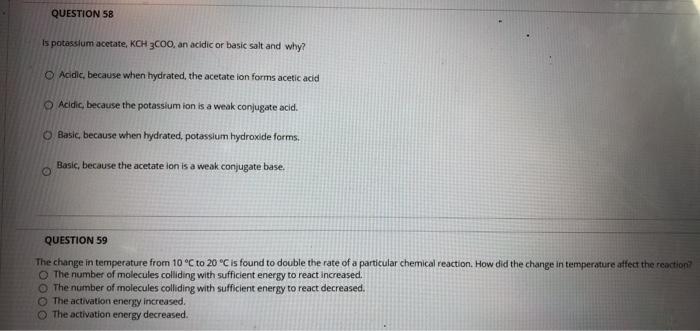
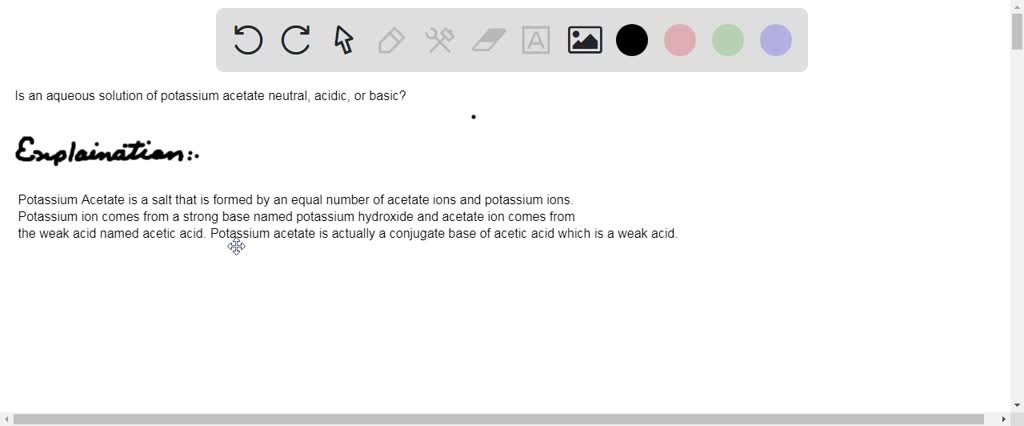
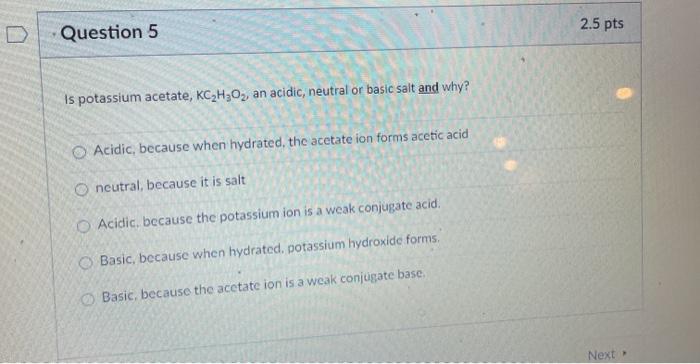
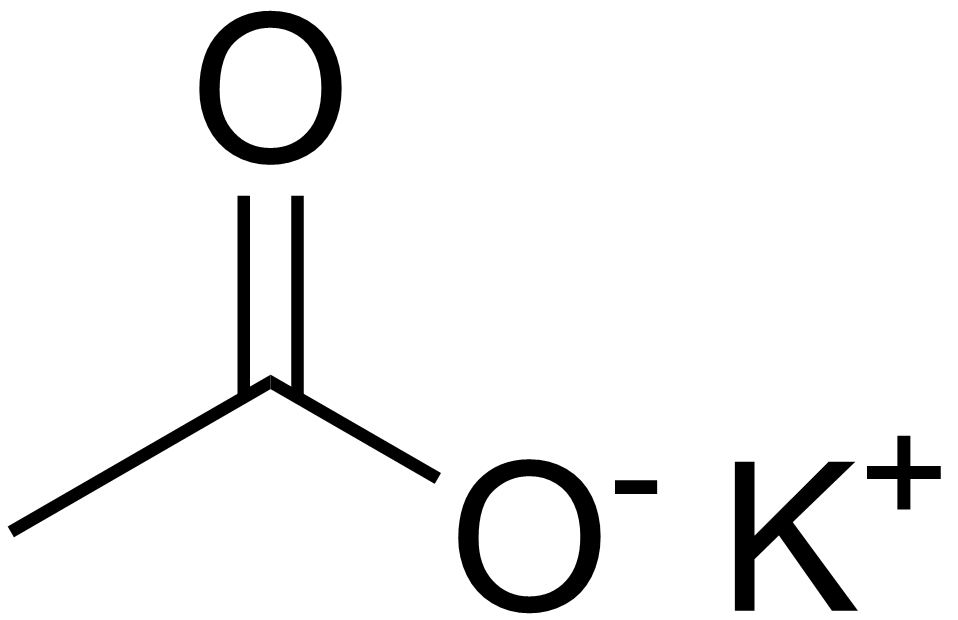
Difference Between Potassium Acetate and Potassium Chloride | Compare the Difference Between Similar Terms

Synthesis of dendralenes 13 and 15: i) potassium acetate, acetic acid,... | Download Scientific Diagram

SOLVED: Which of the following explains why a 0.10 M solution of potassium acetate, KC2H3Oz, is less alkaline (basic) than a 0.10 M solution of potassium cyanide; KCN? Cyanides are less soluble