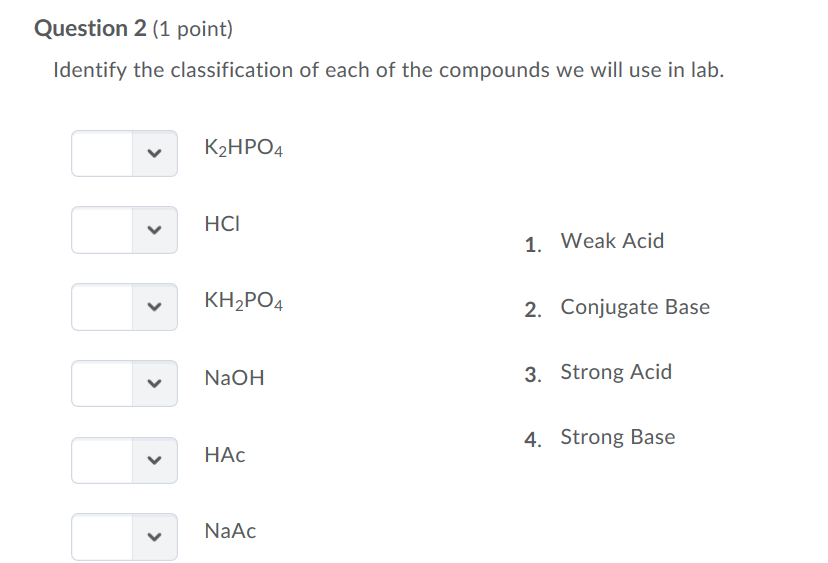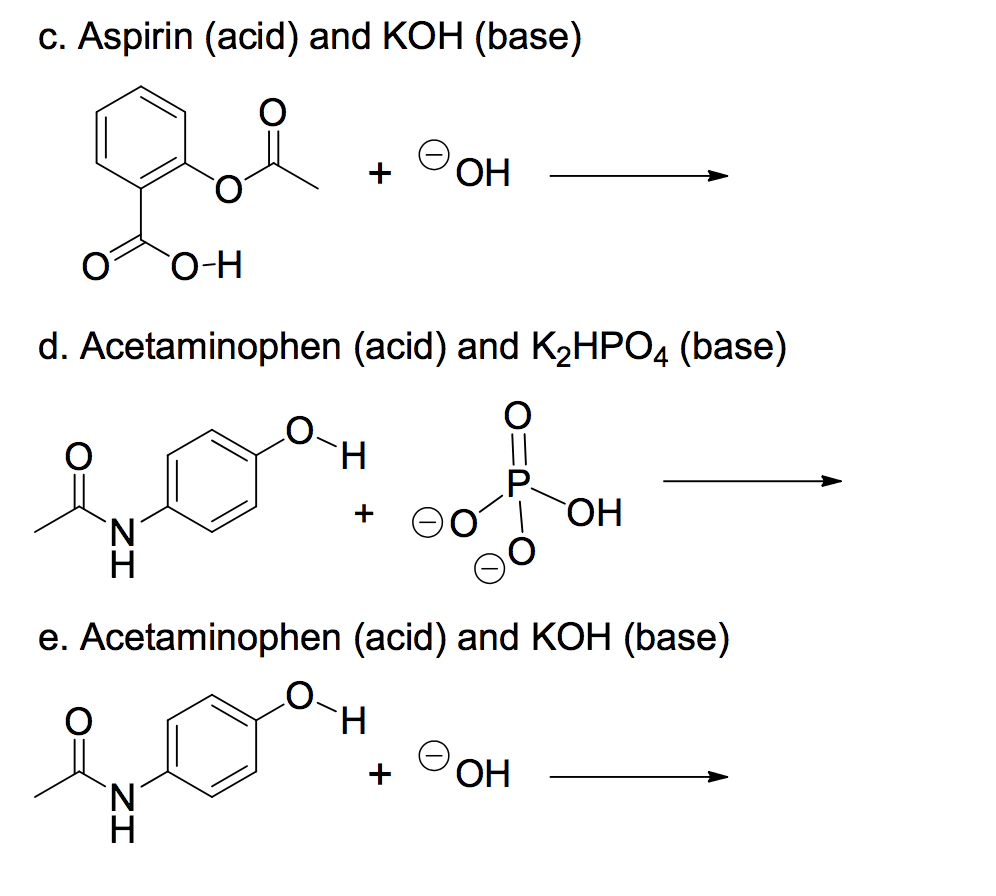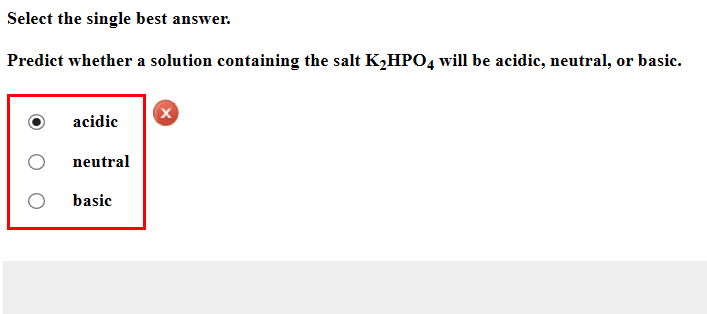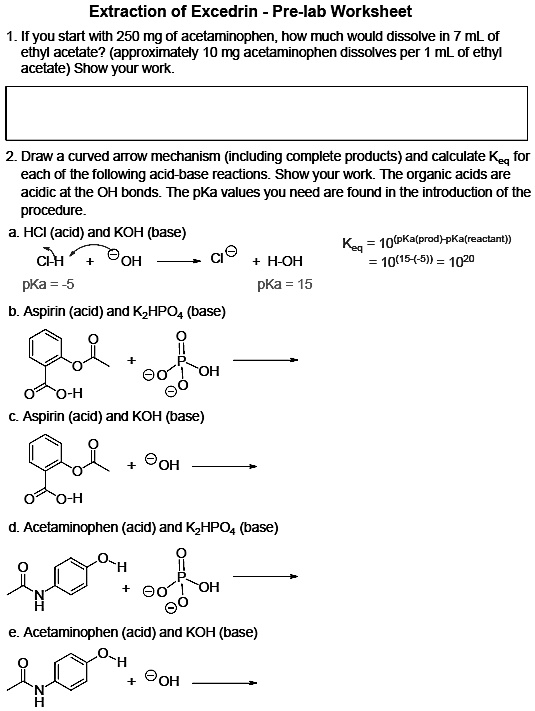
SOLVED: 'I am confused about the pKa and pH when it comes to reacting acid- base chemistry. :( Extraction of Excedrin Pre-lab Worksheet 1. If you start with 250 mg of acetaminophen, how
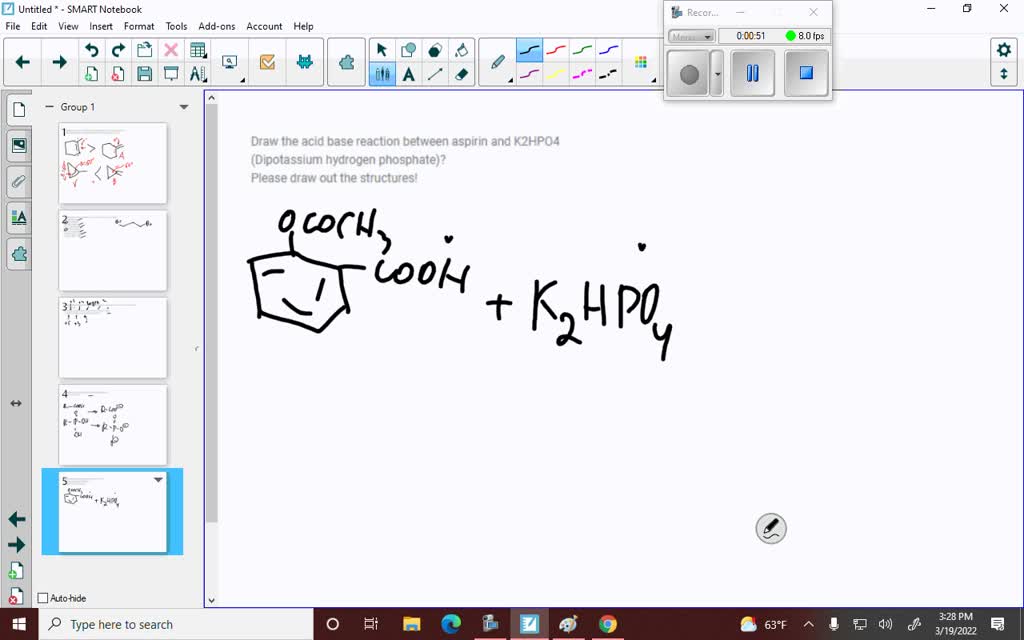
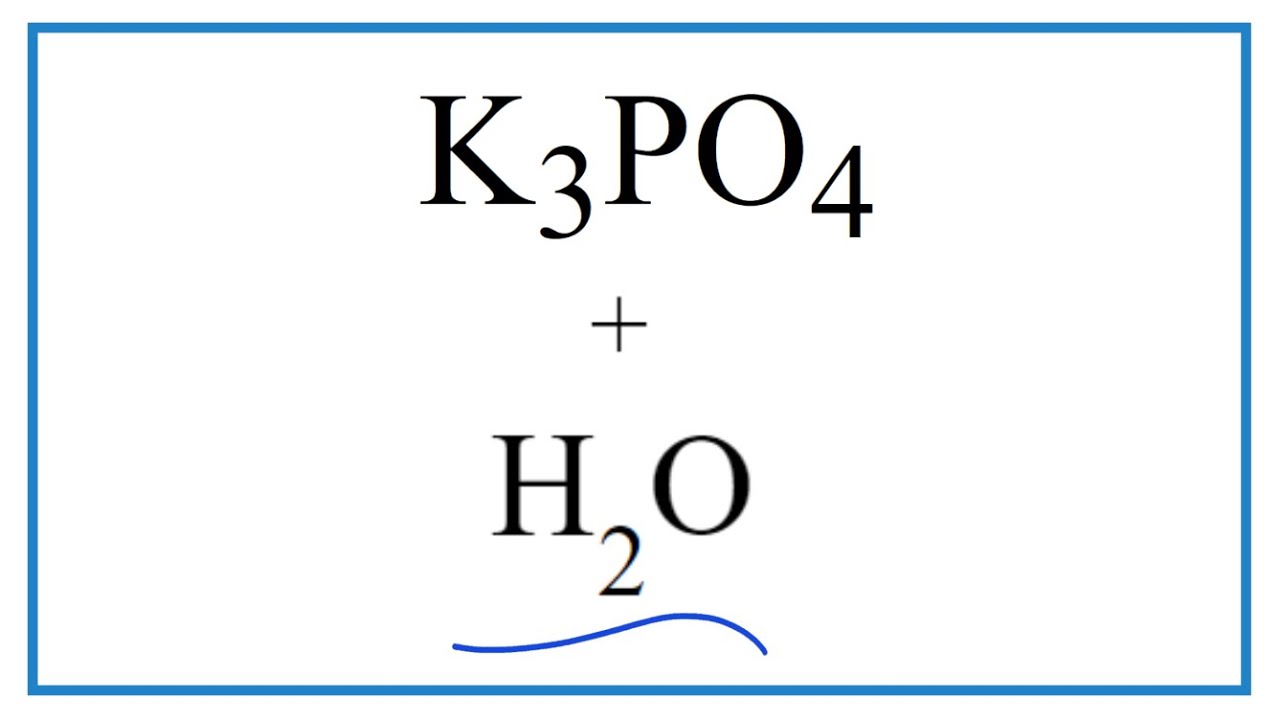
SOLVED: Draw the acid base reaction between aspirin and K2HPO4 (Dipotassium hydrogen phosphate)? Please draw out the structures!