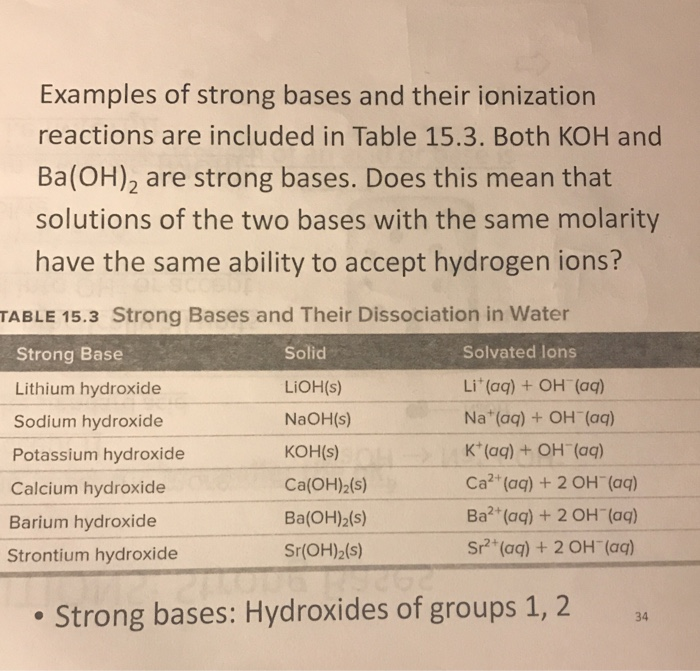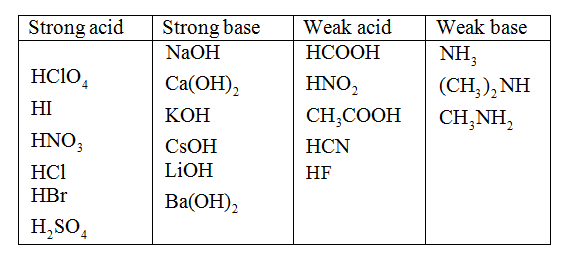
Classify each substance as a strong acid, strong base, weak acid, or weak base - Home Work Help - Learn CBSE Forum
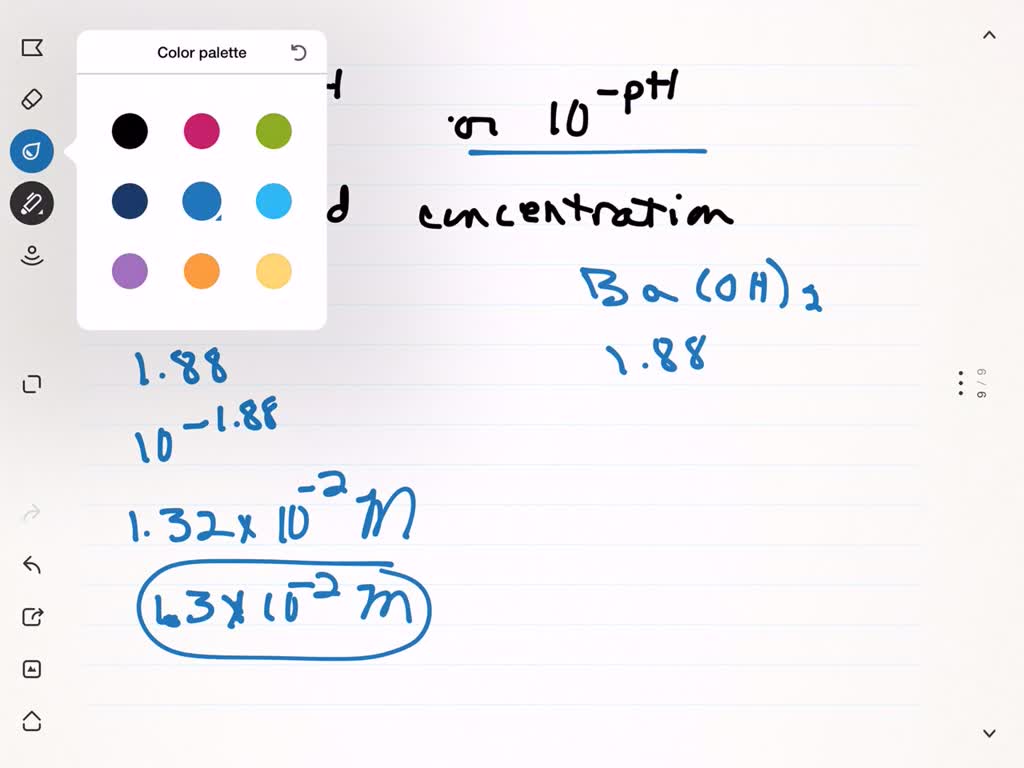
SOLVED:The pOH of a strong base solution is 1.88 at 25^∘ C. Calculate the concentration of the base (a) if the base is KOH and (b) if the base is Ba(OH)2.








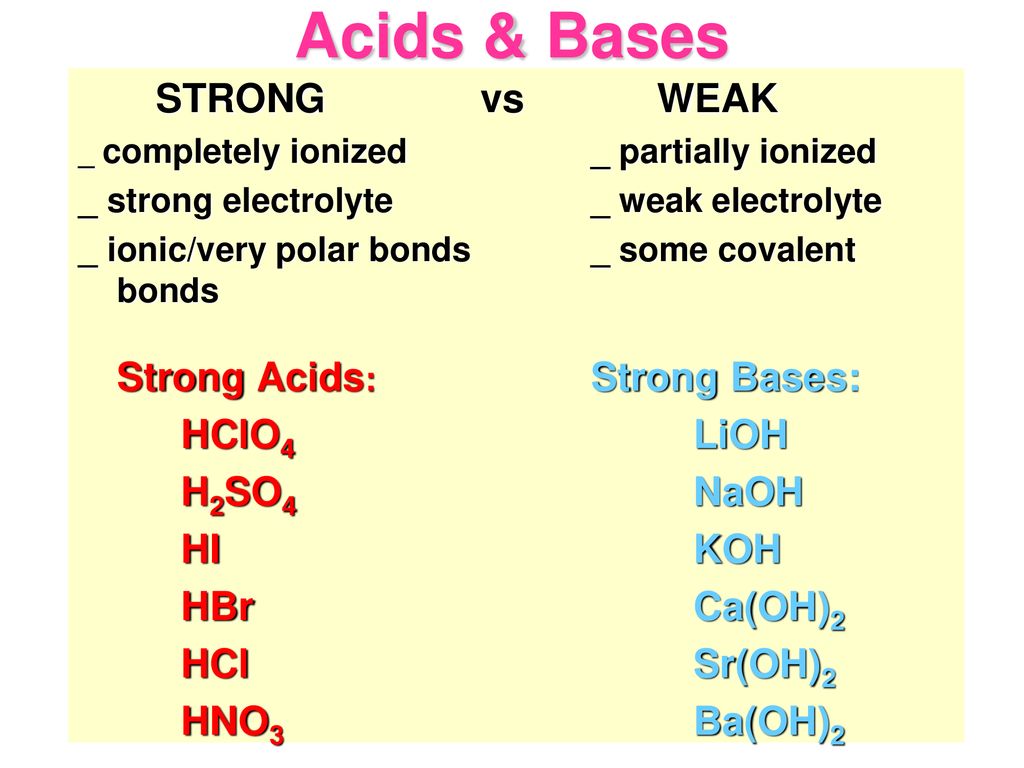





:max_bytes(150000):strip_icc()/most-common-strong-bases-603649-ADD-Final2-a2c0ac3120ff4b65bd98989ee298878c.png)