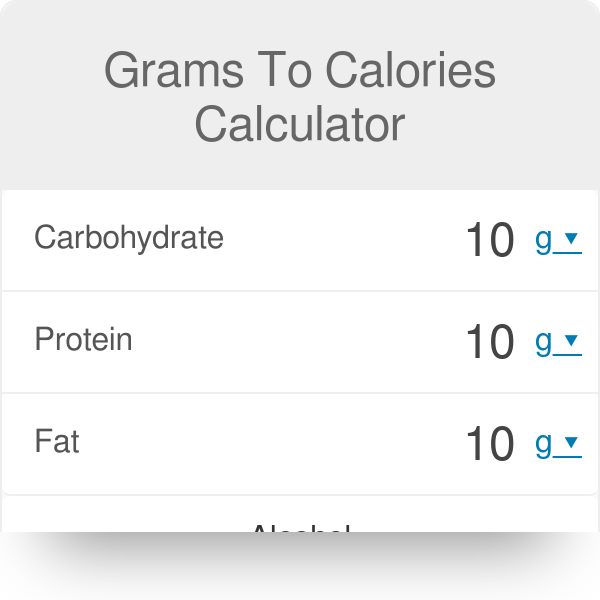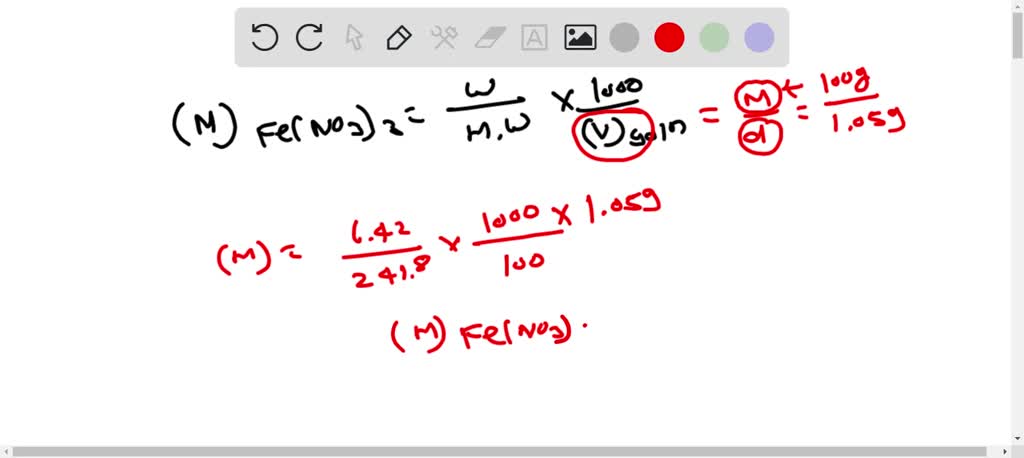
SOLVED: Question 16 Not yet answered 45 g of Ca(NO3)z was used to create a 1.3 M solution: What is the volume of the solution? (molar mass of Ca(NO3)z 164.1 g/mol )

Question Video: Calculating the Mass of Zinc Carbonate that Will Dissolve in 1000 mL of Water | Nagwa
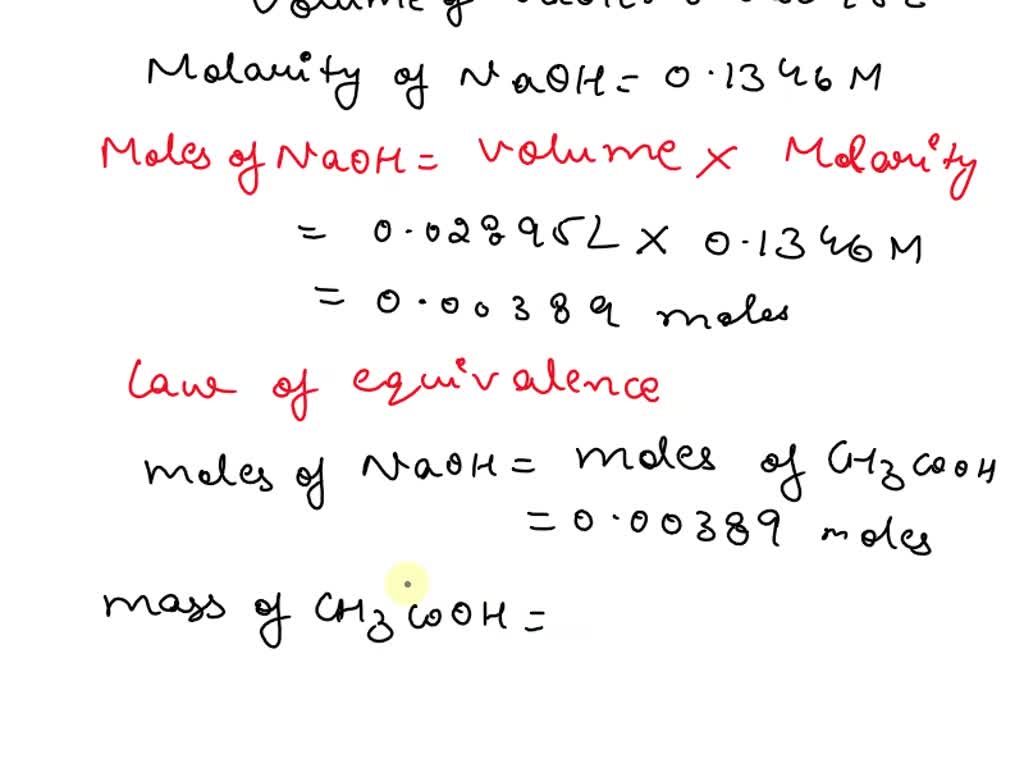
SOLVED: 'Assuming that the density of vinegar is 1.012 g/mL, calculate the weight percent of acetic acid in vinegar if 28.95 mL of 0.1346 M NaOH was required to titrate 5.00 mL of vinegar'

A solution was made by dissolving 3.00 g of Na3PO4 in 50.0 mL of water. The volume of the resulting - YouTube

![Molarity Calculator [with Molar Formula] Molarity Calculator [with Molar Formula]](https://scrn-cdn.omnicalculator.com/chemistry/molarity@2.png)