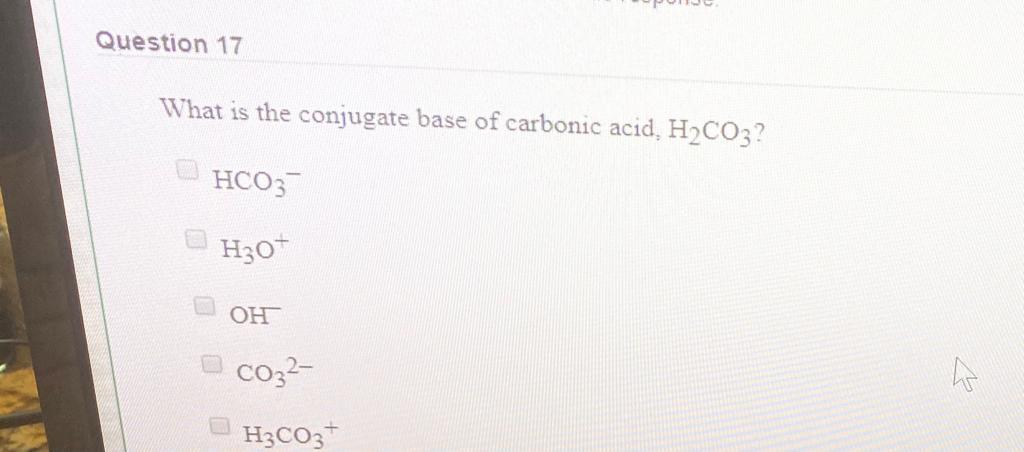Give the conjugate base for each compound below. Acid Conjugate Base H2CO3 h2po-4 nh+4 - Home Work Help - Learn CBSE Forum

SOLVED: Identify the conjugate acid and conjugate base of HCO3" 0 CO32 H2CO32 CO3 H3CO3 HCO32- H2CO3 HCO3

SOLVED: QUESTION 11 In the following reaction: HCO3" (aq) + H2O (aq) -> H2CO3 (aq) + OH" (aq) 0 HCO3" is an acid and is H2CO3 its conjugate base. 0 H20 is
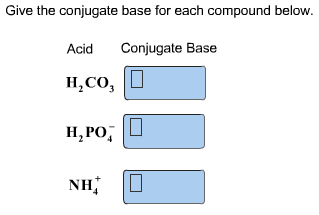
Give the conjugate base for each compound below. Acid Conjugate Base H2CO3 h2po-4 nh+4 - Home Work Help - Learn CBSE Forum