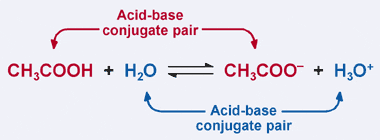
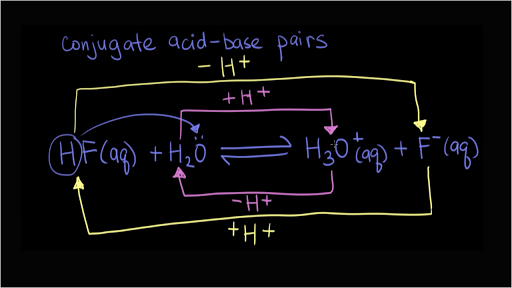
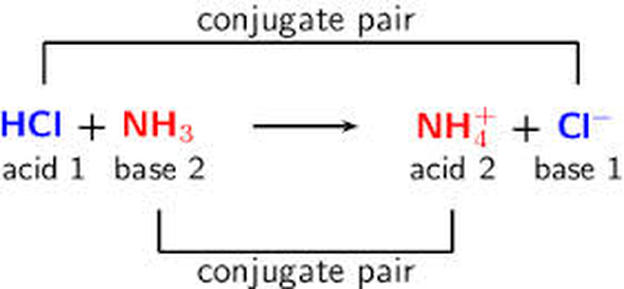
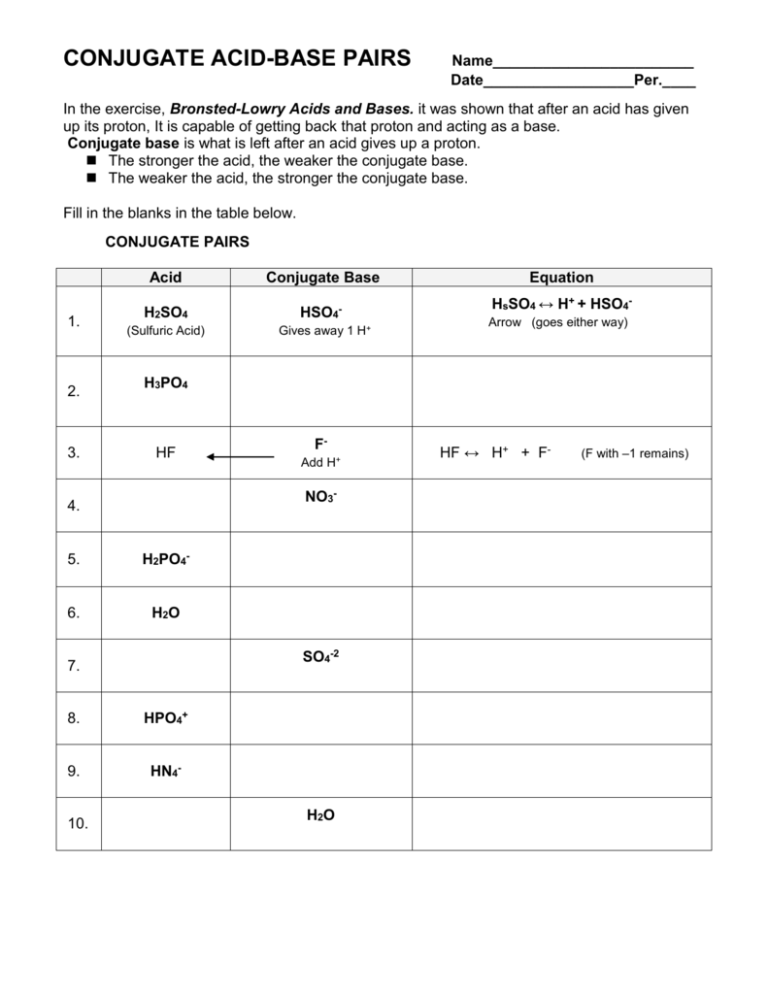
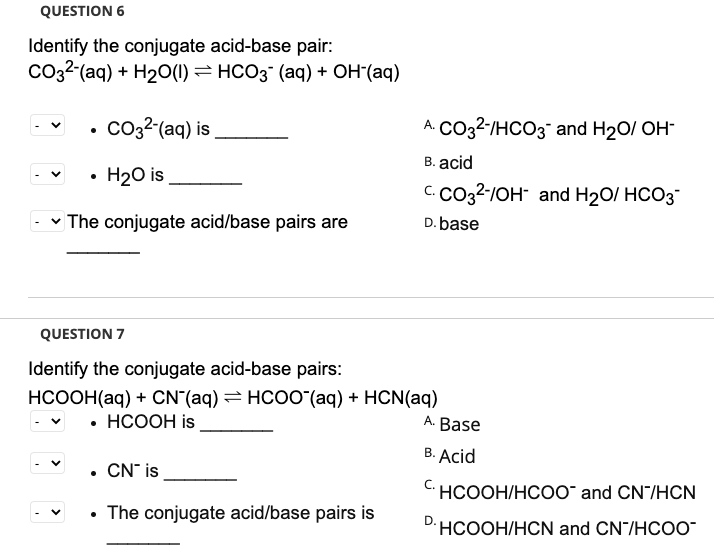
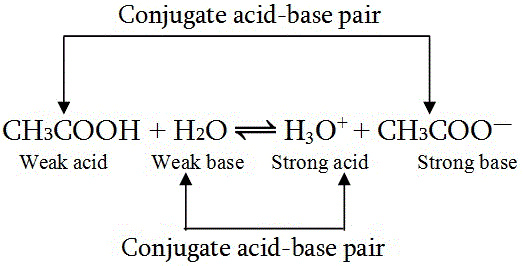
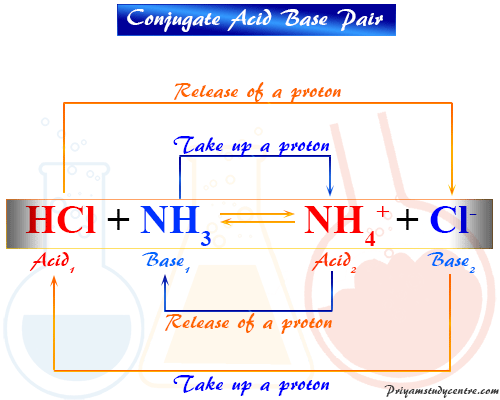
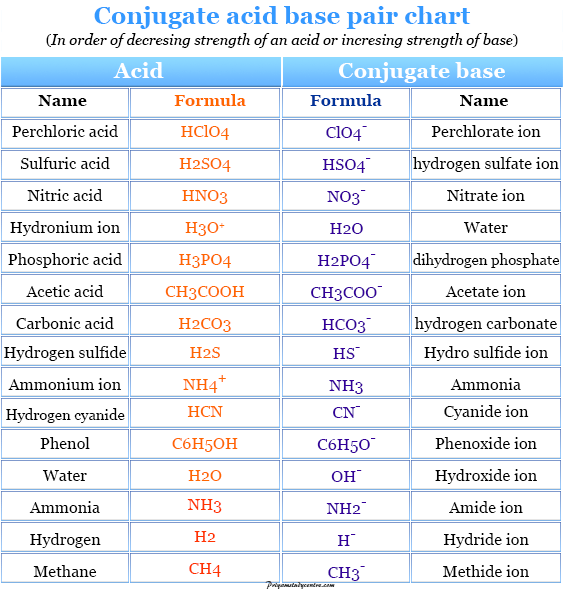
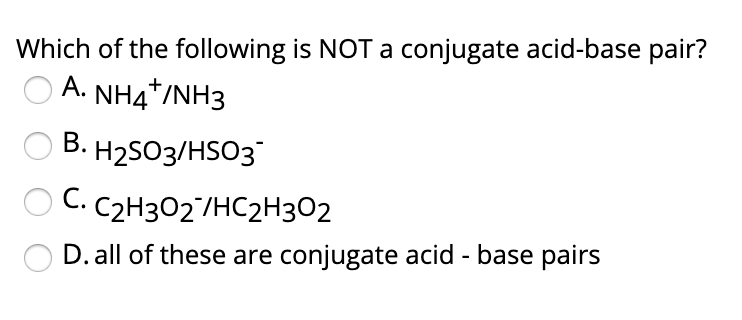SOLVED: 4. Which of the following is a conjugate acid/base pair? (3 Points) H3O+ /OH HCl/OCL - HzSO4/S04 NHt /NH3 noneofthese

Relative strengths of some common conjugate acid- base pairs, listed... | Download Scientific Diagram

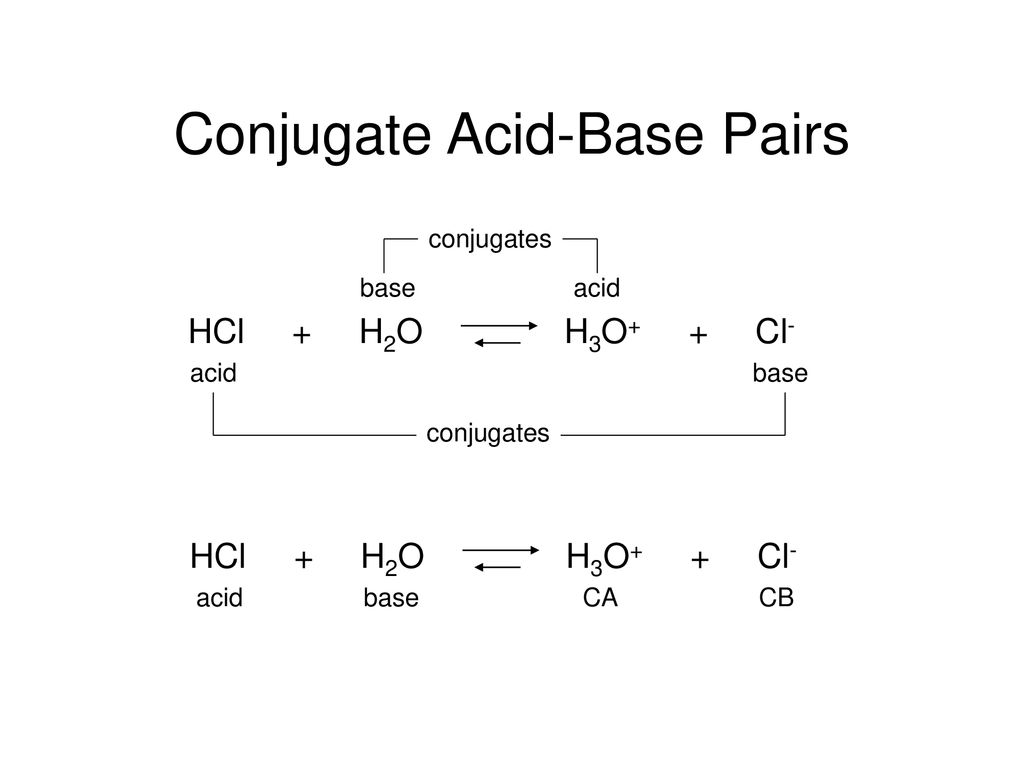
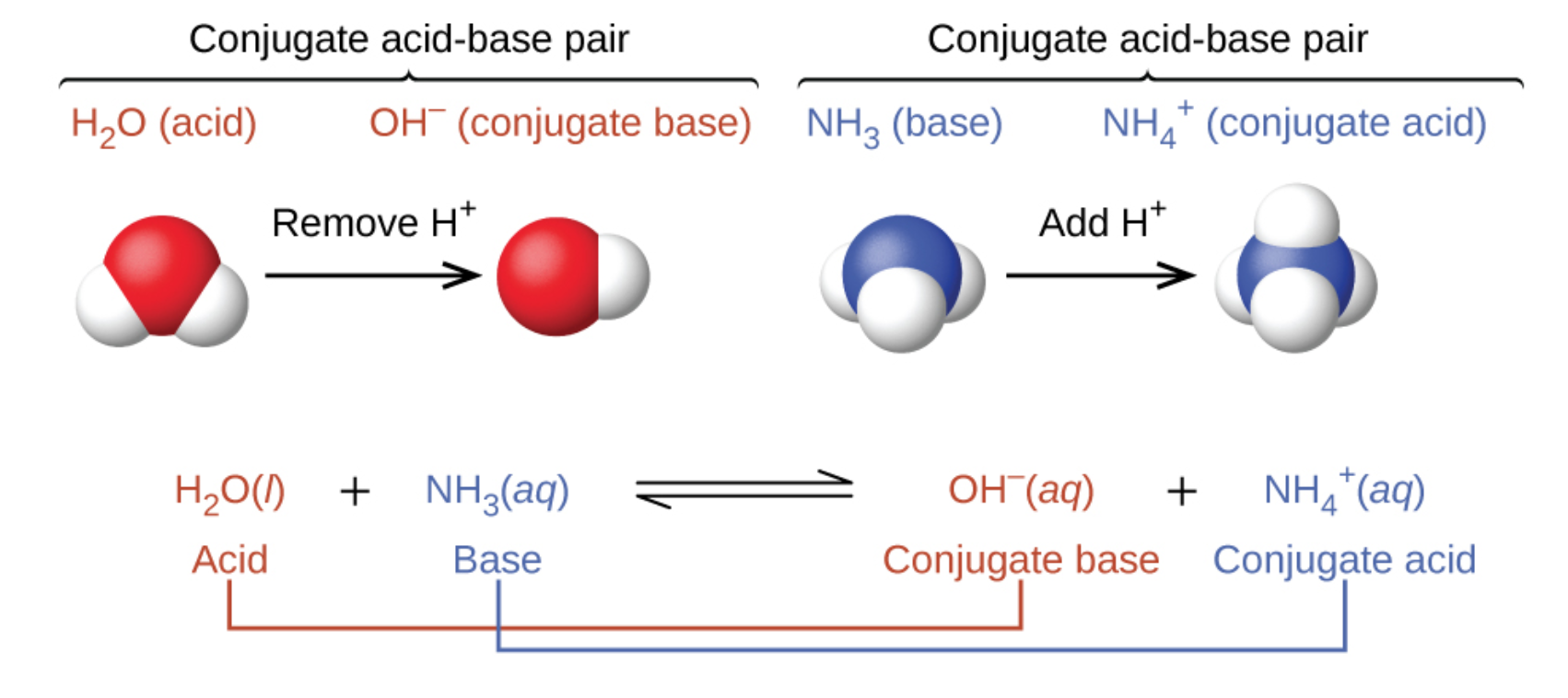
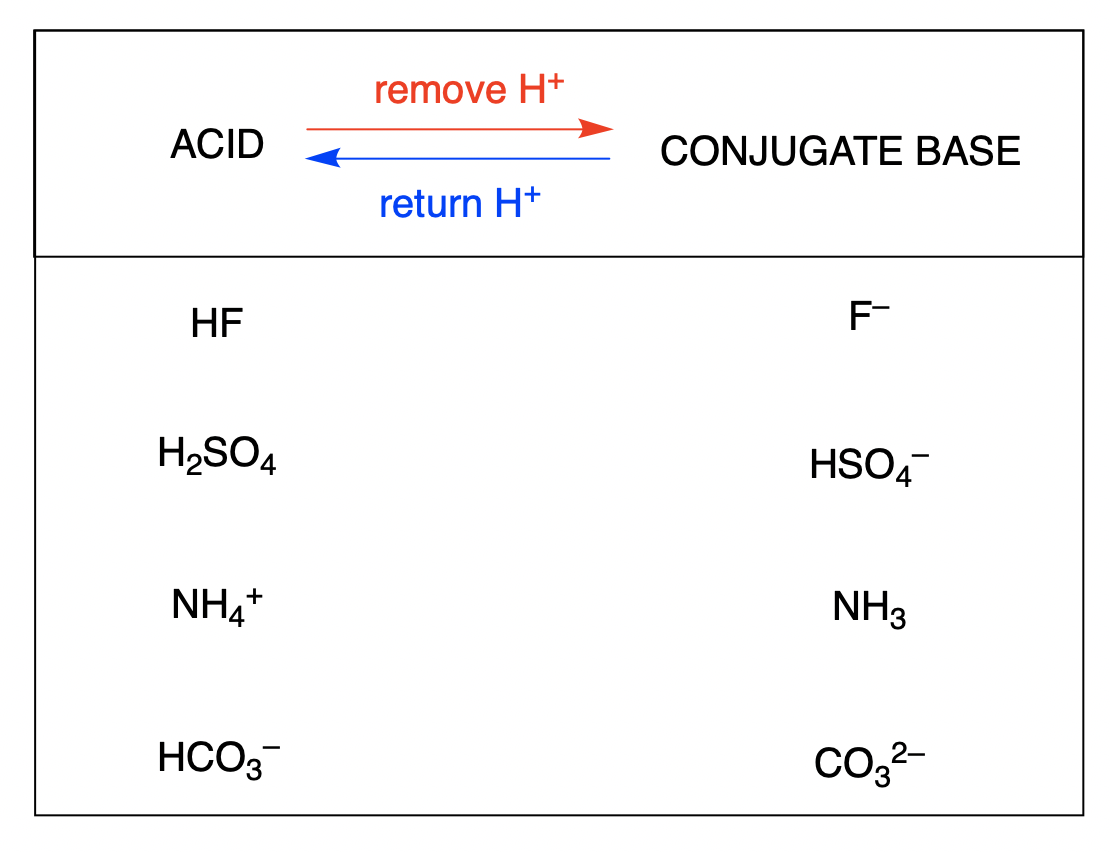
What is meant by the conjugate acid-base pair? Find the conjugate acid/ base for the following species: HNO 2 , CN - , HClO 4 , F - , OH - , CO 3 2- and S 2-

SOLVED: Identify the conjugate acid-base pairs in the following reaction equation: NHzlaq) HCzHaOzlaq) = NHat(aq) + CzH3Oz-(aq) Q25 NHAt/HC2H3Oz and NH3/CzH3Oz NH3/HC2H3Oz and NH4t/C2H302 NH4t/NH3 and HC2H3Oz/C2H3O2 HCzH3Oz/C2H3O2 " is the only